|
3/15/2024 0 Comments Bohrs model of the atom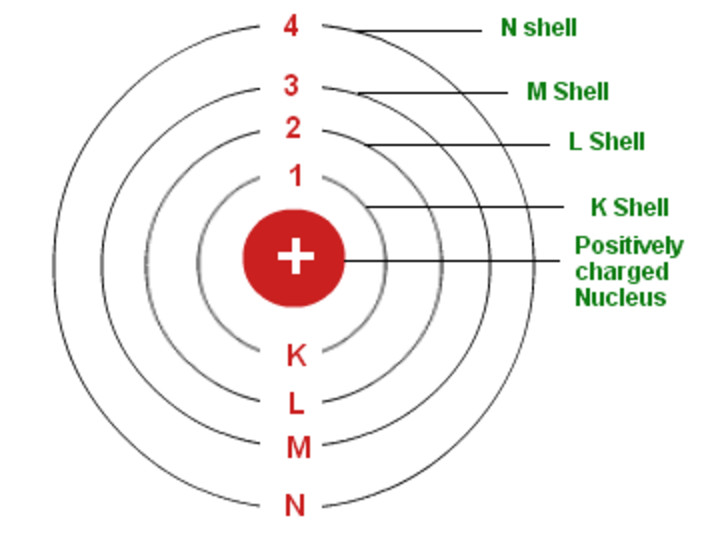
The orbit with n = 1 is the lowest lying and most tightly bound. In his final years, he devoted himself to the peaceful application of atomic physics and to resolving political problems arising from the development of atomic weapons.Īs n decreases, the energy holding the electron and the nucleus together becomes increasingly negative, the radius of the orbit shrinks and more energy is needed to ionize the atom. Bohrs Model was discovered or rather formulated by the Danish physicist Niels Henrik David Bohr. (b) The energy of the orbit becomes increasingly less negative with increasing n.ĭuring the Nazi occupation of Denmark in World War II, Bohr escaped to the United States, where he became associated with the Atomic Energy Project. This was an improvement to the Rutherford model and can be considered as a quantum physical explanation for it. The vast majority of its mass is located in a positively charged central nucleus, which is 10,000. Rutherford had made the startling discovery that most of the atom is empty space. 100th anniversary of Bohrs model of the atom Angew Chem Int Ed Engl. Bohr, one of the pioneers of quantum theory, had taken the atomic model presented a few years earlier by physicist Ernest Rutherford and given it a quantum twist. His enduring insights and superseded suppositions are also discussed. It does introduce several important features of all models used to describe the distribution of electrons in an atom. This Essay traces Bohrs fundamental reasoning regarding atomic structure and spectra, the periodic table of the elements, and chemical bonding. \): The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. Learn about the Bohr model of the atom, a modification of Rutherfords model that explains electrons in fixed orbits around a nucleus. Bohr’s Model of an Atom is a framework comprising a little, thick core encompassed by circling electronslike the structure of the Solar System, yet with fascination given by electrostatic powers instead of gravity. Bohr’s model of the hydrogen atom provides insight into the behavior of matter at the microscopic level, but it is does not account for electronelectron interactions in atoms with more than one electron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
